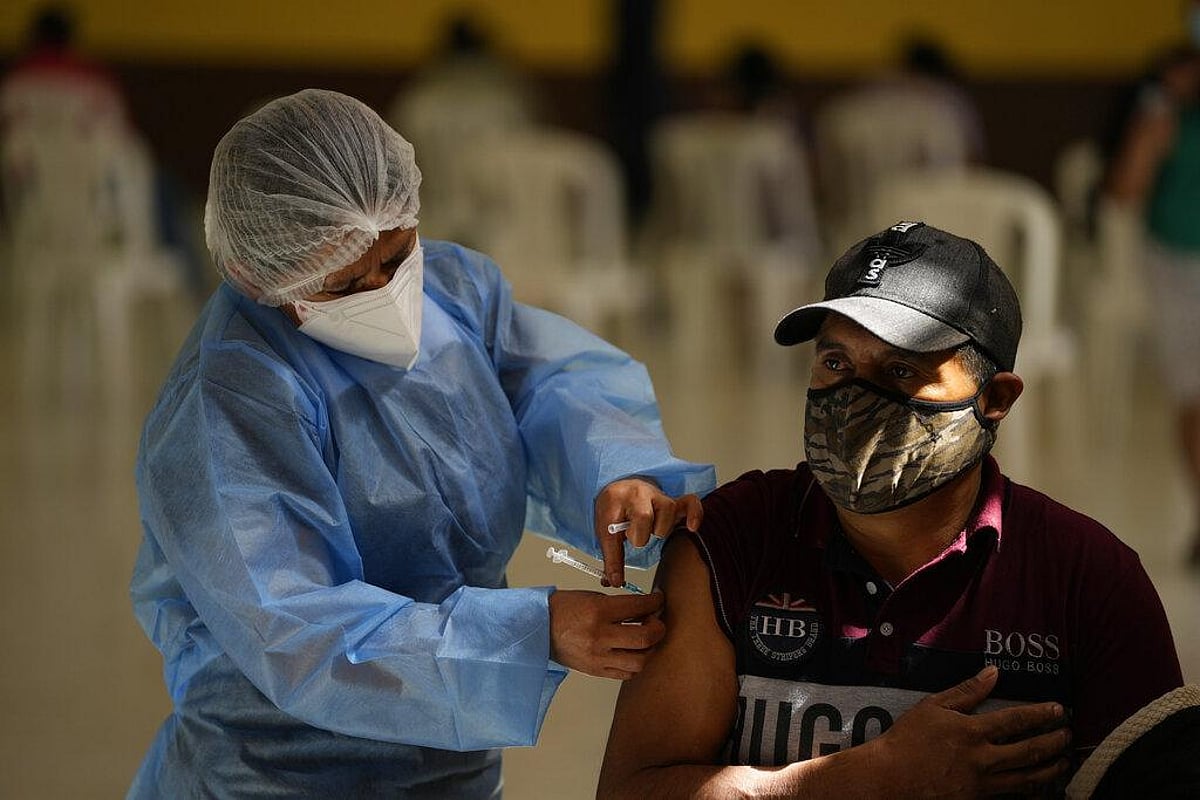
FDA approves Moderna’s next-generation COVID-19 vaccine: Here's what makes it different

Public Health Implications
According to medical experts, this approval is a key milestone in the ongoing COVID-19 mitigation strategy in the US, particularly for vulnerable populations. As community transmission continues to fluctuate, ensuring protection for high-risk individuals remains a public health priority.
However, the CDC is expected to issue formal recommendations regarding rollout logistics and prioritisation in the coming days.
Moderna had in a statement on Saturday said that following the FDA approval, that it remain committed to protecting the most vulnerable populations. "This milestone reflects the strong efficacy and safety data behind our updated platform and reaffirms our role in fighting COVID-19 as it evolves,” it said.
The US vaccine maker also said it is coordinating with federal and state agencies to begin immediate distribution to authorised providers and pharmacies, and the individuals in the approved categories are advised to consult their healthcare provider for eligibility and availability.
Impact in India
As India braces for a potential new epidemic due to the rising number of cases detected across several regions, next-generation vaccines could be particularly relevant in terms of their safety profiles and effectiveness against emerging virus variants.
Advertising by Adpathway